Unlocking Biopharma Innovation With Real-World Evidence
By Partha Anbil, industry advisor, MIT Sloan School of Management, and Anoop Sidharthan

The World Health Organization continues to recognize health data as one of six foundational pillars of effective health systems, alongside service delivery, workforce development, medical products, financing, and governance. However, the volume and velocity of health data generation have increased dramatically since the WHO framework's initial publication. Today, healthcare organizations are generating exponential amounts of structured and unstructured data across EHRs, wearable devices, claims databases, genomic repositories, and patient-reported outcome systems. The challenge has shifted decisively from data scarcity to data integration and intelligent utilization.
In the biopharmaceutical industry, this evolution represents both unprecedented opportunity and significant operational complexity. The 2026 Deloitte Life Sciences Outlook documents that 85% of biopharma executives prioritize investment in data, digital, and AI platforms for research and development, with mature organizations expecting to reduce drug development costs by approximately $1 billion over five-year periods through improved data utilization and adaptive trial design. Simultaneously, regulatory agencies, including the FDA, have issued definitive guidance on incorporating real-world evidence (RWE) into regulatory decision-making. These documents indicate that 25.2% of supplemental approvals for labeling expansions between 2022 and 2024 included RWE, with oncology, infectious diseases, and rheumatology representing the highest-utilization therapeutic areas.
This article discusses how federated data networks, platform aggregators, and collaborative partnerships have evolved to meet the data demands of modern product development, regulatory strategy, and commercial success.
The Evolution of Data Access: 2018 To 2026
The period from 2018 to 2026 brought fundamental shifts in how biopharmaceutical organizations conceptualize and operationalize data access, driven by three converging forces.
- Regulatory Transformation. The FDA's July 2024 release of final guidance on "Real-World Data: Assessing Electronic Health Records and Medical Claims Data to Support Regulatory Decision-Making" formalized an emerging practice. The guidance explicitly addresses the use of EHR data and medical claims data in both interventional and observational studies, providing regulatory certainty that had previously been absent. This formalization accelerated industry adoption, as organizations could now budget and operationalize RWE strategies with confidence in regulatory acceptance.
- Technological Convergence. Advances in cloud infrastructure, federated learning architectures, synthetic data generation, and standard data models have overcome technical barriers that previously constrained multi-organizational data sharing. Modern federated networks can now execute complex analytical queries across geographically dispersed data sets without requiring physical data transfer, addressing privacy concerns that had historically limited collaboration.
- Competitive Necessity. Whereas data access was once a competitive differentiator available primarily to large pharmaceutical companies with substantial resources, it has become a competitive requirement. Smaller biopharmaceutical firms and specialized medical device manufacturers now access RWE through subscription models with aggregator platforms, leveling the competitive landscape.
Stakeholder Priorities and Data Needs
The fundamental stakeholder ecosystem remains consistent with historical frameworks, but clinical research stakeholder priorities have evolved substantially.
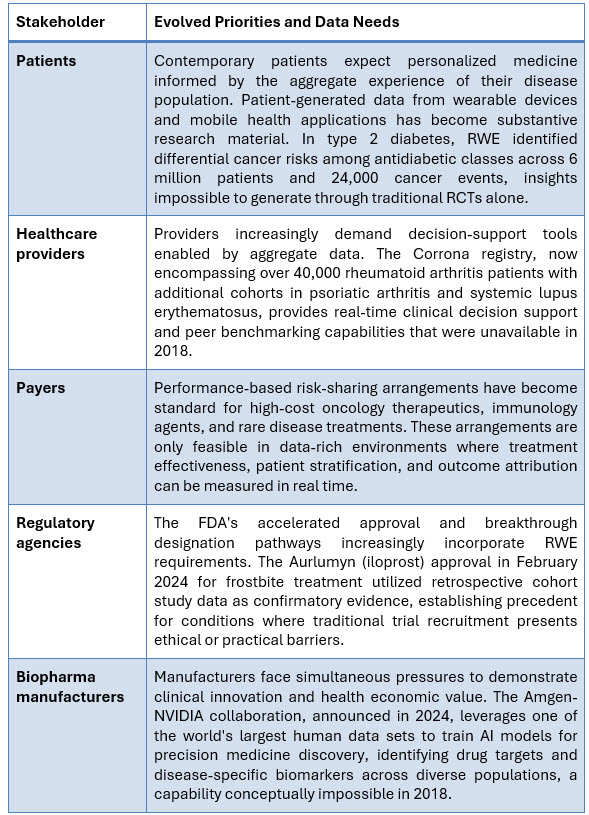
Therapeutic Area Case Studies
Oncology: Precision Medicine Through RWE. Oncology represents the therapeutic area with the most advanced data access implementation, with 43.6% of FDA labeling expansion approvals incorporating RWE between 2022 and 2024. Flatiron Health, acquired by Roche in 2018, transforms EHRs from hundreds of cancer treatment facilities into research-grade databases, enabling comparative effectiveness analysis across treatment protocols and correlations between biomarkers and treatment outcomes. A landmark example: RWD analysis showed that HER2-low breast cancer patients, previously considered ineligible for HER2-targeted therapy, demonstrated meaningful benefit from trastuzumab deruxtecan, leading to an expanded FDA label and fundamentally changing treatment algorithms in 2024. Additionally, Vijoice (alpelisib) received FDA approval in April 2022 based substantially on a single-arm study using medical record data from seven sites across five countries.
Rheumatology: Multi-Indication Registries. Corrona has expanded from a rheumatoid arthritis focus to operate parallel registries for psoriatic arthritis, ankylosing spondylitis, systemic lupus erythematosus, and multiple sclerosis. Contemporary innovations include integration of genomic data (HLA-B27 status, ACPA/RF antibody titers, genetic polymorphisms affecting drug metabolism) with clinical phenotyping and treatment response, enabling subpopulation analyses that drive both improved clinical outcomes and valuable regulatory evidence for label expansions into newly characterized patient subgroups.
Cardiology: Risk Stratification and Prevention. Large-scale RWE studies have established risk-stratification algorithms that predict cardiovascular events across diverse populations. A 2024 RWE analysis of 1.2 million patients across integrated health systems demonstrated that GLP-1 receptor agonist therapy reduces the risk of cardiovascular events and certain obesity-associated cancers, expanding utilization patterns and prompting healthcare systems to develop protocols for earlier initiation of GLP-1 therapy in high-risk patients. The evidence translated directly into changes to treatment algorithms affecting hundreds of thousands of patients.
Endocrinology/Metabolism. RWE-driven treatment algorithms have revolutionized type 2 diabetes management. Integration of claims data, clinical data, and patient-reported data has enabled the development of sophisticated algorithms predicting which medication class best serves individual patient needs. The discovery that SGLT-2 inhibitors offer protective effects against specific cancer types emerged not from hypothesis-driven trials but from systematic analysis of RWD linking medication exposure to cancer outcomes across millions of patients, exemplifying insights that would require decades of prospective follow-up but become available through intelligent analysis of existing databases.
Infectious Disease. The PEDSnet federated network, which aggregates medical records from pediatric healthcare systems, enabled the collection of safety data on new dosing regimens through RWE, accelerating regulatory decision-making for Vimpat (lacosamide) pediatric seizure management compared with traditional trial approaches.
Rare Disease. The Achondroplasia Natural History registry provided external control data for the approval of Voxzogo (vosoritide) in November 2021, enabling regulatory review where traditional RCTs would require multiyear recruitment at substantial cost. This pattern has become standard: Patient advocacy organizations develop registries, biopharmaceutical companies subsidize data collection, and regulatory agencies accept registry-derived external controls in approval pathways.
Contemporary Data Access Models
Federated query networks have evolved from experimental approaches to the industry standard by 2026. These networks, where data remains with original custodians and analytical queries are distributed rather than centralized, address privacy concerns while enabling large-scale analysis. The Nordic Federated Health initiative (2024-2026) demonstrates international federated implementation, linking EHRs across Nordic countries to identify adverse drug reactions and improve patient safety. Contemporary federated architecture typically includes distributed data nodes, common data model mapping, query service interfaces, federated learning algorithms, synthetic data generation capabilities, and robust governance structures addressing privacy, security, and data use authorization.
Platform aggregators have emerged for specific therapeutic domains. In oncology, platforms such as CancerIQ and Syapse combine genomic data, treatment protocols, and biomarker information within oncology-specific data models. In rheumatology, Corrona integrates biomarker data, imaging assessments, patient-reported outcomes, and healthcare utilization patterns, with participating providers accessing real-time decision support that compares their patients' outcomes with network benchmarks. For infectious diseases, platforms tracking antimicrobial resistance and pathogen genomics have become integral to antibiotic stewardship.
Nonprofit and academic data custodianship, exemplified by the Yale University Open Data Access (YODA) model, has become increasingly important. Academic institutions serving as trusted intermediaries create structures where independent parties control data access and publication decisions, addressing the tension between manufacturers' evidence needs and scientific independence. By 2026, multiple academic health systems manage data-custody functions, enabling independent researchers to access proprietary data sets through formal data-use agreements.
RWE platforms have evolved from data vendors to intelligence partners, providing therapeutic area-specific expertise, regulatory strategy guidance, comparative effectiveness analysis frameworks, and market access support. This evolution reflects recognition that data access alone provides limited value; interpretation, contextualization, and strategic application create actual value.
Conditions For Success and Barriers
Organizations successfully leveraging data access by 2026 possess robust cloud-based analytics infrastructure, common data model capability, interoperability protocols (FHIR, HL7, DICOM), and synthetic data infrastructure. They also maintain clear governance frameworks including data stewardship protocols, privacy compliance across jurisdictions (HIPAA, GDPR, LGPD, PIPL), institutional review board engagement, and transparent data use agreements. Successful implementations further demonstrate patient involvement, provider partnership, transparent communication, and equitable benefit distribution.
Despite these advances, significant barriers persist. Healthcare data remains fragmented across incompatible systems. International regulatory complexity adds substantial cost. Vendor consolidation has raised concerns about monopolistic control over data access, though public investment in federated networks (FDA Sentinel, PCORnet) partially mitigates this. RWE often reflects historical patterns of healthcare access stratified by race, ethnicity, and socioeconomic status, risking the perpetuation of inequities if not explicitly addressed through diversity analyses. Analytical complexity and methodological inconsistency across studies, including selective reporting and inadequate accounting for unmeasured confounding, remain familiar challenges.
Strategic Imperatives For 2026 And Beyond
Organizations must prioritize investment in data science talent, as advanced data scientists, biostatisticians, and clinical informatics specialists have become the limiting resource. Development of therapeutic area expertise is essential, because generic data access capabilities provide limited value without deep clinical knowledge. Commitment to pragmatic research design ensures analyses answer questions relevant to clinical practice. Patient-centered analytics explicitly incorporate patient priorities in research question development. The ultimate vision of real-time learning health systems, which continuously adjust treatment protocols based on emerging evidence, remains largely aspirational but increasingly feasible.
Conclusion
Data access has evolved from a strategic advantage to an operational necessity in contemporary biopharmaceutical and medical device development. The federated networks, regulatory acceptance of RWE, and multi-stakeholder collaborations that were emerging in 2018 have matured into standard practice by 2026. For biopharmaceutical executives, the imperative is no longer whether to prioritize data access but how to implement it effectively, through simultaneous investment in technical infrastructure, analytical talent, and governance structures. The 25% of labeling expansions incorporating RWE between 2022 and 2024, combined with FDA guidance providing clear expectations, suggests this proportion will continue to increase. Organizations that have already implemented RWE infrastructure maintain a competitive advantage; organizations beginning this journey in 2026 face elevated barriers but can still access platforms and expertise to support rapid implementation. The ultimate measure of success is not technical elegance or regulatory acceptance but improved patient outcomes. Data access initiatives that genuinely improve clinical decision-making, enable earlier disease diagnosis, and accelerate the discovery of novel therapeutic targets create enduring value for patients, and this patient-centered orientation should guide all investment decisions.
Authors’ Note: The views expressed in the article are those of the authors and not of the organizations they represent.
About The Authors:
 Partha Anbil is at the intersection of the life sciences industry and management consulting. He is currently SVP, Life Sciences, at Coforge Limited, a $1.7 billion multinational digital solutions and technology consulting services company. He held senior leadership roles at WNS, IBM, Booz & Company, Symphony, IQVIA, KPMG Consulting, and PWC. Anbil has consulted with and counseled health and life sciences clients on structuring solutions to address strategic, operational, and organizational challenges. He was a member of the IBM Industry Academy, a highly selective group of professionals inducted by invitation only, the highest honor at IBM. He is a healthcare expert member of the World Economic Forum (WEF). He is also a life sciences industry advisor at MIT, his alma mater.
Partha Anbil is at the intersection of the life sciences industry and management consulting. He is currently SVP, Life Sciences, at Coforge Limited, a $1.7 billion multinational digital solutions and technology consulting services company. He held senior leadership roles at WNS, IBM, Booz & Company, Symphony, IQVIA, KPMG Consulting, and PWC. Anbil has consulted with and counseled health and life sciences clients on structuring solutions to address strategic, operational, and organizational challenges. He was a member of the IBM Industry Academy, a highly selective group of professionals inducted by invitation only, the highest honor at IBM. He is a healthcare expert member of the World Economic Forum (WEF). He is also a life sciences industry advisor at MIT, his alma mater.
 Anoop Sidharthan is practitioner of enterprise technology architecture and digital transformation with deep specialization in cloud, generative AI, and healthcare & life sciences. He brings experience spanning architecture leadership roles at Cognizant and L&T Infotech. Sidharthan has architected and delivered large-scale transformation programs exceeding $500 million, counseling global enterprises on cloud modernization, multi-cloud strategy, and AI-driven solutions across payer, provider, life sciences, and pharma domains. He has led technology implementations across the USA, U.K., Australia, and India and is a trusted advisor to business and IT stakeholders on enterprise architecture and healthcare ecosystem integration.
Anoop Sidharthan is practitioner of enterprise technology architecture and digital transformation with deep specialization in cloud, generative AI, and healthcare & life sciences. He brings experience spanning architecture leadership roles at Cognizant and L&T Infotech. Sidharthan has architected and delivered large-scale transformation programs exceeding $500 million, counseling global enterprises on cloud modernization, multi-cloud strategy, and AI-driven solutions across payer, provider, life sciences, and pharma domains. He has led technology implementations across the USA, U.K., Australia, and India and is a trusted advisor to business and IT stakeholders on enterprise architecture and healthcare ecosystem integration.
